 These are the conditions under which most reactions are carried out in the laboratory the system is usually open to the atmosphere (constant pressure) and we begin and end the process at room temperature (after any heat we have added or which is liberated by the reaction has dissipated. Remember that \(ΔG\) is meaningful only for changes in which the temperature and pressure remain constant. This subtlety is discussed in a bit more detail in a footnote in Kerson Huang's book in page 16.\), which this equation expresses in an indirect way, is the only criterion of spontaneous change. As with any other state function, the change in entropy is defined as the difference between the entropies of the final and initial states: S S f S i. How can entropy, S, be a state function if it has different values at states 1 and 2 depending on if the process is reversible or irreversible i. This means that the net change in entropy during a complete cycle is zero, so that entropy is a function of state. Then we will derive the entropy formula for ideal gas, ' S(N V E) NkB ln 4 mE 3Nh2 32 + 5 from the microcanonical (NV E) ensemble. Where $\delta Q_$ by a reversible process. 10 In this lecture, we will rst discuss the relation between entropy and irreversibility. In fact, the Clausius statement of the 2nd law says this: ItmaybehelpfultoseethatifweuseGibbsentropyde nitionthatentropydoesinfactincrease duringdi usion.Forsimplicityconsideradi usinggasin1dimension,withnumberdensityn(x t). We know that the entropy transferred to a system undergoing a reversible cycle is zero, and if you have intuition behind this, then you have intuition behind why entropy is a state function. In order to quantify this, let us consider another simple thought experiment. As observed, Fermis aim to explain entropy (not only its mathematical expression) is reduced to the definition of a state function without concern for its. Thus, any process that gives rise to an increase in the number of microscopic states in a system will also lead to an increase in the entropy. Now, since $S=k_Bln\Omega$, entropy is also a state function.ĮDIT: I've given some intuition behind the statistical mechanical definition of entropy being a state function, but what about the thermodynamic definition? We noted that entropy is a thermodynamic state function related to the number of microscopic states available to a system. There is no such a thing as an amount of work or heat in a system. Other quantities such as work ((w)) and heat ((q)), on the other hand, are not state functions. State functions depend only on the state of the system. Therefore $\Omega=\Omega(N,P,T.)$ is a state function. All this is irrelevant to specify the pressure, entropy, etc, because all these variables are what we call state functions. Intuitively, you can think of the number of accessible energy states by particles in a system being influenced by the state of the system (e.g. It should be added that, even before a proper definition of entropy was posited, entropy was confirmed as a state function, which means that whenever the.  
When a gas expands into a vacuum, its entropy increases because the increased volume allows for greater atomic or molecular disorder.  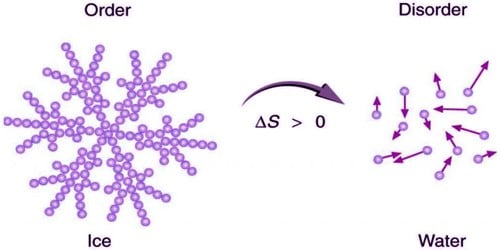
Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other thermodynamic. The number of available energy levels is determined by quantities like temperature, which is also influenced by pressure and other thermodynamic parameters. As with any other state function, the change in entropy is defined as the difference between the entropies of the final and initial states: S S f S i. Entropy is a state function that is often erroneously referred to as the state of disorder of a system. The number of microstates is determined by the number of particles (of course) and the number of energy levels available to these particles. Entropy is proportional to the number of microstates available to a system, $S=k_Bln\Omega$. Theoriginalde nitionofentropy,duetoClausius,wasthermodynamic.Aswesawinthelast lecture,Clausiusnotedthatentropyisafunctionofstate,wecancalculatetheentropydi erence betweentwostatesbyconnectingthemhoweverwelike. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
